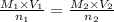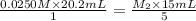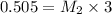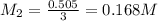Ferris & mona used the orp sensor to titrate a ferrous ammonium sulfate solution, (nh4)2fe(so4)2 with kmno4 titrant.
they titrated a 15.00 ml aliquot of the fe+2 solution with 0.0250 m mno4- solution and determined that the equivalence point was at 20.2 ml.
what is the molarity of the fe+2 solution? 5 fe+2(aq) + mno4-(aq) + 8 h+(aq) â 5 fe+3(aq) + mn+2(aq) + 4 h2oselect one: a. 0.168 mb. 0.0928 mc. 0.0337 md. 0.673 m

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:00, WinterStrikesBack
Solutions of sodium carbonate and silver nitrate react to form solid silver carbonate and a solution of sodium nitrate. a solution containing 3.50 g of sodium carbonate is mixed with one containing 5.00 g of silver nitrate. how many grams of sodium carbonate, silver nitrate, silver carbonate, and sodium nitrate are present after the reaction is complete?
Answers: 2

Chemistry, 22.06.2019 14:50, ladybugperez05
Which of the following is most likely true about water in chemical systems? a) water dissolves nonpolar ionic compounds. b) water dissociates ionic compounds. c) water dissociates covalent molecules. d) water dissolves nonpolar covalent substances.
Answers: 1

Chemistry, 22.06.2019 18:30, bibiansolis
The table lists the lattice energies of some compounds. compoundlattice energy (kj/mol)lif –1,036licl –853naf –923kf –821nacl –786which statement about crystal lattice energy is best supported by the information in the table? the lattice energy increases as cations get smaller, as shown by lif and kf. the lattice energy increases as the cations get larger, as shown by lif and licl. the lattice energy decreases as cations get smaller, as shown by nacl and naf. the lattice energy decreases as the cations get smaller, as shown by naf and kf.
Answers: 3
You know the right answer?
Ferris & mona used the orp sensor to titrate a ferrous ammonium sulfate solution, (nh4)2fe(so4)...
Questions in other subjects:


Mathematics, 20.09.2020 18:01


Chemistry, 20.09.2020 18:01






Business, 20.09.2020 18:01