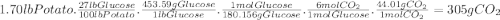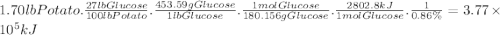Chemistry, 22.11.2019 04:31 mclendenen8011
Photosynthesis allows plants to turn light energy into chemical energy by forming glucose from carbon dioxide and water: 6co2(g) + 6h2o(l) → c6h12o6(g) + o2(g) δhrxn = 2802.8 kj the glucose is then used to form complex carbohydrates that the plant needs to survive. a 1.70 lb sweet potato is approximately 73% water by mass. if the remaining mass is made up of carbohydrates derived from glucose (mw = 180.156 g/mol), how much carbon dioxide (mw = 44.01 g/mol) was needed to grow this sweet potato? g how much light energy does it take to grow the 1.70 lb. sweet potato if the efficiency of photosynthesis is 0.86%? kj

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 09:30, janetexcoelho
What does the mass of 0.7891 mol of ferric oxide (fe2o3)
Answers: 1

Chemistry, 22.06.2019 11:00, coco8560
Freezing and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1

Chemistry, 22.06.2019 14:00, luisaareli6298
Calculate the energy required to ionize a hydrogen atom to an excited state where the electron is initially in the n = 5 energy level. report your answer in kilojoules
Answers: 1
You know the right answer?
Photosynthesis allows plants to turn light energy into chemical energy by forming glucose from carbo...
Questions in other subjects:


Mathematics, 14.12.2020 19:30


Mathematics, 14.12.2020 19:30

Chemistry, 14.12.2020 19:30

Business, 14.12.2020 19:30