
Chemistry, 21.11.2019 20:31 ilovewaffles70
Which statement best explains why the polarity of a h2o molecule differs from the polarity of a co2 molecule? the bond strength is greater in co2. oxygen is more electronegative than carbon. the central atom in h2o has lone pair electrons and the central atom in co2 does not.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:30, sotoamerica0814
98 ! and brainliest plz ! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 22.06.2019 10:40, yfgkeyonna
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2

Chemistry, 22.06.2019 13:20, alejandra340
Can someone me with 3 and 4 plz. this is for masteries test.
Answers: 2

Chemistry, 22.06.2019 17:20, alexis3060
How do you know when a chemical reaction has occurred
Answers: 1
You know the right answer?
Which statement best explains why the polarity of a h2o molecule differs from the polarity of a co2...
Questions in other subjects:

Mathematics, 24.02.2021 04:20

History, 24.02.2021 04:20


Mathematics, 24.02.2021 04:20



Chemistry, 24.02.2021 04:20

Mathematics, 24.02.2021 04:20

Mathematics, 24.02.2021 04:20

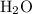 molecule differs from the polarity of
molecule differs from the polarity of 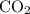 molecule, because Oxygen is more electronegative than carbon.
molecule, because Oxygen is more electronegative than carbon. is 3.44.
is 3.44.


