
Chemistry, 21.11.2019 03:31 briannamaee13
Areaction was performed in which 0.55 g of 2‑naphthol was reacted with a slight excess of allyl bromide to make 0.59 g of allyl 2‑naphthyl ether. calculate the theoretical yield and percent yield for this reaction. allyl bromide has a density of 1.40 g/ml .

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:10, andersonemma2222
How do you identify the anode on a power source such as a battery? how do you identify the cathode? how are terms anion and cation?
Answers: 1

Chemistry, 22.06.2019 22:00, jespinozagarcia805
In order to complete this lab. you will need to be familiar with some common chemistry terms. complete the chemical change puzzle and list the relevant terms and their meaning below a. rectant b. product c. supernate
Answers: 3

Chemistry, 23.06.2019 00:00, samangelzrose3576
Exit what is the density of an object having a mass of 5.0 g and a volume of 45.0 cm3?
Answers: 1

Chemistry, 23.06.2019 12:30, okasiafolk27
15) a substance used in manufacturing gasoline consists of finely divided platinum supported on an inert solid. suppose that the platinum is formed by the high temperature reaction between platinum (iv) oxide and hydrogen gas. the other product is water. a) write and balance the equation b) how many grams of hydrogen are needed to produce 1.0 g of platinum metal? c) how many moles of water are produced at the same time? how many grams? ( show work, .)
Answers: 1
You know the right answer?
Areaction was performed in which 0.55 g of 2‑naphthol was reacted with a slight excess of allyl brom...
Questions in other subjects:



Mathematics, 23.03.2021 18:40




Mathematics, 23.03.2021 18:40


Mathematics, 23.03.2021 18:40

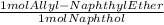 * 184 gEther/mol = 0.7028 g
* 184 gEther/mol = 0.7028 g


