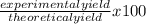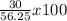Chemistry, 20.11.2019 23:31 jacobs5919
If 39.0 g of c6h6 reacts with excess chlorine and produces 30.0 g of c6h5cl in the reaction c6h6 + cl2 → c6h5cl + hcl , what is the percent yield of c6h5cl? 1. 50.0% 2. 53.4% 3. 69.4% 4. 13.2% 5. 76.9%

Answers: 2


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 12:20, missayers172
Achemistry student weighs out 0.306 g of citric acid (h3c6h5o7), a triprotic acid, into a 250 ml volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with 0.1000 m naoh solution. calculate the volume of naoh solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.
Answers: 3
You know the right answer?
If 39.0 g of c6h6 reacts with excess chlorine and produces 30.0 g of c6h5cl in the reaction c6h6 + c...
Questions in other subjects:

Social Studies, 19.04.2021 22:00

Mathematics, 19.04.2021 22:00

Chemistry, 19.04.2021 22:00


Mathematics, 19.04.2021 22:00



Geography, 19.04.2021 22:00