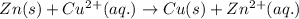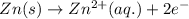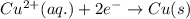Chemistry, 20.11.2019 17:31 fowers8376
Which species functions as the oxidizing agent in the following reduction-oxidation reaction:
zn(s) + cu^2+(aq) > cu(s) + zn^2+(aq)
a) zn^2+(aq)
b) zn(s)
c) cu^2+
d) cu(s)

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:40, sugardime
Choose an equation that represents an enzyme-catalyzed reaction. (a) enzyme + substrate → enzyme-substrate complex (b) enzyme + substrate ←−→ enzyme + products (c) enzyme + substrate ←−→ enzyme-substrate complex → enzyme + products (d) enzyme + substrate ←−→ enzyme-substrate complex → enzyme-substrate complex + products
Answers: 2

Chemistry, 22.06.2019 19:20, halledoll2002
Anyone who's in connections academy chemistry b have the factors that affect the rate of a reaction portfolio already done?
Answers: 3

Chemistry, 22.06.2019 22:00, huddyxo
Scientists often have to deal with numbers that are either very large or very small. for example, the radius of the sun is approximately 696,000 kilometers, while bacterial cells are as small as 1.9 × 10-4 millimeters. express each number in an alternate form.
Answers: 1
You know the right answer?
Which species functions as the oxidizing agent in the following reduction-oxidation reaction:
Questions in other subjects:




History, 20.11.2019 20:31






Chemistry, 20.11.2019 20:31

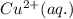 is the oxidizing agent for the given equation.
is the oxidizing agent for the given equation.