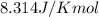Chemistry, 19.11.2019 23:31 jadenjohnson89
Match the following1. a measurement of the volume that the gas occupies.2. a measure of the total amount of the gas.3. a measurement of the absolute.4. a number representing the universal. 5. a value representing the absolute. a. usually measured in liters. b. usually measured in moles. c.pressure of a gas. d.gas constant. e.temperature.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:00, jmanrules200
What is the empirical formula of vanadium 1 oxide given that 20.38 grams of vandium combines with oxygen to form 23.58 grams of the oxide
Answers: 1

Chemistry, 21.06.2019 23:30, mastershadow2018
Agroup of students is studying convection currents. they fill two identical balloons with the same amount of helium. one balloon is placed in a freezer and the other in an area with warm air. after 10 minutes, the balloons are released from a height of 1 meter. which of the following do the students most likely observe? a. the balloons both rise. the cold balloon is larger than the warm balloon. b. the balloons rise at the same rate. both balloons are the same size. c. the warm balloon expands and rises. the cold balloon shrinks and sinks. d. the cold balloon expands and rises. the warm balloon shrinks and sinks.
Answers: 2

Chemistry, 22.06.2019 04:40, deedee363
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3
You know the right answer?
Match the following1. a measurement of the volume that the gas occupies.2. a measure of the total am...
Questions in other subjects:



Biology, 22.09.2019 20:50




English, 22.09.2019 20:50

Biology, 22.09.2019 20:50

History, 22.09.2019 20:50

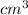 ,
, 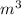 , L and ml.
, L and ml.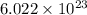 .
.