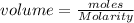a. moles = molarity × volume.

All of the statements about molarity are correct except :
a. moles = molarity × volume.
b. the molarity of a diluted solution is less than the molarity of the original solution.
c. the abbreviation is m. volume = moles/molarity.
d. the interpretation of the symbol is "moles of solute per mole of solvent."

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 09:00, Ezekielcassese
What is the percentage composition of carbon in the compound ch4
Answers: 1

Chemistry, 22.06.2019 14:50, rebeccamckellpidge
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 22.06.2019 17:00, Estrella2209
Which property of a rock remains unchanged by mechanical weathering? a. total surface area b. size and shape c. mineral composition d. sharpness
Answers: 1
You know the right answer?
All of the statements about molarity are correct except :
a. moles = molarity × volume.
a. moles = molarity × volume.
Questions in other subjects:

Chemistry, 09.11.2020 18:10


Chemistry, 09.11.2020 18:10

Mathematics, 09.11.2020 18:10


Chemistry, 09.11.2020 18:10

English, 09.11.2020 18:10



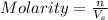
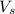 = volume of solution in L
= volume of solution in L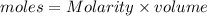
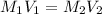
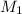 = molarity of stock solution
= molarity of stock solution 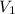 = volume of stock solution
= volume of stock solution 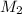 = molarity of diluted solution
= molarity of diluted solution 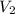 = volume of diluted solution
= volume of diluted solution