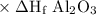Calculate the standard enthalpy of the reaction, δh∘rxn, for the thermite reaction:
2al(s)+fe2o3(s)→2fe(s)+al2o3(s)
elements in their standard state have an enthalpy of formation value of zero. the standard enthalpies of formation of fe2o3 and al2o3 are
δh∘f of fe2o3(s)=−825.5 kj/molδh∘f of al2o3(s)=−1675 kj/mol
express your answer to three significant figures and include the appropriate units.

Answers: 1


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 08:00, kleathers97
Nconcentration refers to the molar concentration of an ion in solution. it may be identical to, or greater or less than, the molar concentration of the compound containing the ion that was used to make the solution. for soluble salts, the molarity of a particular ion is equal to the molarity of that compound times the subscript for that ion. for example, 1 m of alcl3 is 1 m in al3+ and 3 m in cl−. 1 m of (nh4)2so4 is 2 m in nh4+ and 1 m in so42−. part a what is the concentration of k+ in 0.15 m of k2s? view available hint(s) nothing m m part b if cacl2 is dissolved in water, what can be said about the concentration of the ca2+ ion? view available hint(s) if is dissolved in water, what can be said about the concentration of the ion? it has the same concentration as the cl− ion. its concentration is half that of the cl− ion. its concentration is twice that of the cl− ion. its concentration is one-third that of the cl− ion. part c a scientist wants to make a solution of tribasic sodium phosphate, na3po4, for a laboratory experiment. how many grams of na3po4 will be needed to produce 550 ml of a solution that has a concentration of na+ ions of 0.700 m ? express your answer numerically in grams. view available hint(s) mass of na3po4 n a 3 p o 4 = nothing g provide feedback
Answers: 3

Chemistry, 22.06.2019 10:30, freddhendrickss
When the speed of the bottle is 2 m/s, the average maximum height of the beanbag is m.
Answers: 2
You know the right answer?
Calculate the standard enthalpy of the reaction, δh∘rxn, for the thermite reaction:
2al(s)+fe...
2al(s)+fe...
Questions in other subjects:


Chemistry, 14.01.2021 21:00

English, 14.01.2021 21:00



Social Studies, 14.01.2021 21:00


English, 14.01.2021 21:00


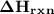 ) can be given by:
) can be given by:
 + 1
+ 1 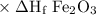
 + 1
+ 1