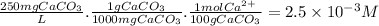The buffer solution is used to control the ph to insure that it does not become too high because excessively basic solutions could cause the corresponding hydroxides of hard metal ions (such as ca(oh)2 and mg(oh)2) to precipitate. using the calcium ion as a typical representative, just how high a ph do you think could be considered as "too high" for a solution with a hardness of about 250 ppm caco3? ksp for ca(oh)2 is 6.5 x 10-6.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:10, shafferakr6
There are 6.022 x 10^23 atoms of hg in 1 mole of hg. the number of atoms in 4.5 moles of hg can be found by multiplying 4.5 by 6.022 x 10^23 a. 2.7 x 10^24 b. 27 x 10^23 c. 2.71 x10^24 d. 27.099 x 10^23
Answers: 3

Chemistry, 22.06.2019 22:10, zwbaby3693
Which aqueous solution of ki freezes at the lowest temperature? 1) 1 mol of ki in 500. g of water 2) 2 mol of ki in 500. g of water 3) 1 mol of ki in 1000. g of water 4) 2 mol of ki in 1000. g of water
Answers: 3


Chemistry, 23.06.2019 14:00, Gaby702
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 6.00 mol fe and 8.45 mol nio(oh) react?
Answers: 1
You know the right answer?
The buffer solution is used to control the ph to insure that it does not become too high because exc...
Questions in other subjects:



Mathematics, 19.01.2022 14:00



English, 19.01.2022 14:00

SAT, 19.01.2022 14:00

SAT, 19.01.2022 14:00


SAT, 19.01.2022 14:00