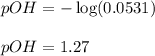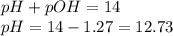Chemistry, 15.11.2019 07:31 accioanswers
An analytical chemist is titrating of a solution of cyanic acid with a solution of . the of cyanic acid is . calculate the ph of the acid solution after the chemist has added of the solution to it. note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of solution added.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:20, lindseysmith9522
Neils bohr believed that electrons orbited the nucleus in different energy levels, based on strong support from
Answers: 1


Chemistry, 23.06.2019 01:30, jarteria0
Some molecular compounds, such as hcl, ionize completely in solution. for molecular compounds such as h2co3, most molecules do not ionize in solution. which describes the properties of these two solutes? a. hcl and h2co3 have the same effect on the properties of the solution. b. hcl raises the freezing point of water more than h2co3 does. c. hcl raises the boiling point of water more than h2co3 does.
Answers: 2

Chemistry, 23.06.2019 07:40, Aaron5795
)in the deacon process for the manufacture of chlorine, hcl and o2 react to form cl2 and h2o. sufficient air (21 mole% o2, 79% n2) is fed to provide 35% excess oxygen, and the fractional conversion of hcl is 85%. calculate the mole fractions of the product stream components.
Answers: 1
You know the right answer?
An analytical chemist is titrating of a solution of cyanic acid with a solution of . the of cyanic a...
Questions in other subjects:

Mathematics, 01.02.2020 20:42



History, 01.02.2020 20:42


Biology, 01.02.2020 20:42




Mathematics, 01.02.2020 20:42

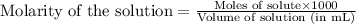 .....(1)
.....(1)
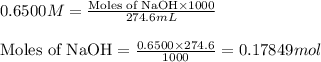
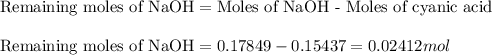
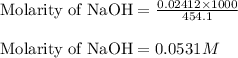
![pOH=-\log[OH^-]](/tpl/images/0375/6220/fe336.png)
![[OH^-]=0.0531M](/tpl/images/0375/6220/f41e8.png)