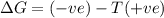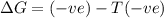Chemistry, 15.11.2019 02:31 williejaroid123
Classify the possible combinations of signs for a reaction's δh and δs values by the resulting spontaneity
a. δh is positive and δs is negative
b. δh is positive and δs is positive
c. δh is negative and δs is positive
d. δh negative and δs is negative
for a, b, c and d find out which of following they are:
1. spontaneous as written at all temperatures
2. spontaneous in reverse at all temperatures
3. spontaneous as written above a certain temperature
4. spontaneous as written below a certain temperature
they can only have one answer but 2 can have the same answer

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:00, wizz4865
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2


Chemistry, 22.06.2019 13:30, princessroseee769
Ants live on acacia trees in south america. the ants feed on sugars secreted by the trees. the trees provide room for the ants to live. the ants sting any other insect or animal that comes to eat the trees. what type of relationship is this?
Answers: 1

Chemistry, 22.06.2019 15:30, vivianfling
Why does earth rotate? because earth is formed from cold gases collapsing due to gravity because the matter in the nebula that formed earth was spinning because earth forms more than 99% of the mass of the solar system because the hydrogen atoms inside the nebula fused to form helium
Answers: 1
You know the right answer?
Classify the possible combinations of signs for a reaction's δh and δs values by the resulting spont...
Questions in other subjects:

Mathematics, 15.11.2019 21:31

Social Studies, 15.11.2019 21:31

History, 15.11.2019 21:31





Mathematics, 15.11.2019 21:31

Spanish, 15.11.2019 21:31

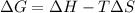
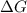 = Gibbs free energy
= Gibbs free energy 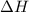 = enthalpy change
= enthalpy change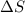 = entropy change
= entropy change 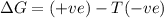
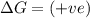
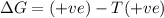
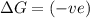 (at high temperature) (spontaneous)
(at high temperature) (spontaneous)