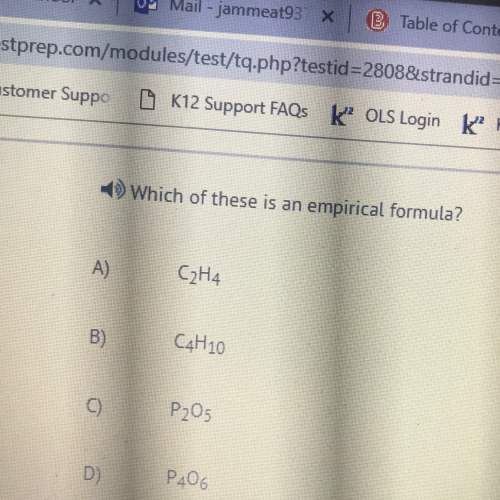
A35.6 g sample of ethanol (c2h5oh) is burned in a bomb calorimeter, according to the following reaction. if the temperature rose from 35.0 to 76.0 c and the heat capacity of the calorimeter is 23.3 kj/ c, what is the value of dh rxn? the molar mass of ethanol is 46.07 g/mol.
c2h5oh(l) + o2(g) --> co2(g) + h2o(g) ? h_ rxn = ?
a) -1.24 * 103 kj/mol
b) +1.24 * 103 kj/mol
c) -8.09 * 103 kj/mol
d) -9.55 * 103 kj/mol
e) +9.55 * 103 kj/mol

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:40, neonbluefaith
Which statement best describes the function of enzymes?
Answers: 1

Chemistry, 22.06.2019 18:00, liddopiink1
An object displaces 652 ml of water. the volume of the object is: 0.652 cm³ 6.52 cm³ 65.2 cm³ 652 cm³
Answers: 3

Chemistry, 22.06.2019 19:10, asdfghhk9805
How does the atmosphere to make earth livable? check all that apply. causes the seasons contains oxygen provides warmth creates important nutrients blocks harmful energy from the sun plz like !
Answers: 2

Chemistry, 22.06.2019 21:30, djdjdjdbdbjx
What is another way to determine mass times acceleration?
Answers: 1
You know the right answer?
A35.6 g sample of ethanol (c2h5oh) is burned in a bomb calorimeter, according to the following react...
Questions in other subjects:








Mathematics, 22.06.2019 22:00

Mathematics, 22.06.2019 22:00