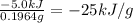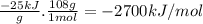Chemistry, 13.11.2019 21:31 joooosshhhh
Quinine is an important type of molecule that is involved in photosynthesis. the transport of electrons mediated by quinone in certain enzymes allows plants to take water, carbon dioxide, and the energy of sunlight to create glucose. a 0.1964-g sample of quinone (c6h4o2) is burned in a bomb calorimeter with a heat capacity of 1.56kj/c. the temperature of the calorimeter increases by 3.2 degrees c. calculate the energy of combustion of quinone per gram and per mole.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:50, kelli151
An aqueous solution of potassium hydroxide is standardized by titration with a 0.194 m solution of hydrobromic acid. if 25.2 ml of base are required to neutralize 24.2 ml of the acid, what is the molarity of the potassium hydroxide solution? m potassium hydroxide
Answers: 2


Chemistry, 22.06.2019 23:00, NewKidnewlessons
What is the energy in joules of a mole of photons associated with visible light of wavelength 486 nm?
Answers: 3

Chemistry, 23.06.2019 03:00, draveon6925
Achemical equilibrium between gaseous reactants and products is shown. n2(g) + 3h2(g) ⇌ 2nh3(g) how will the reaction be affected if the pressure on the system is increased? it will shift toward the reactant side as there is lower pressure on the reactant side. it will shift toward the product side as there is higher pressure on the product side. it will shift toward the reactant side as there are a greater number of moles of gas on the reactant side. it will shift toward the product side as there are a fewer number of moles of gas on the product side.
Answers: 2
You know the right answer?
Quinine is an important type of molecule that is involved in photosynthesis. the transport of electr...
Questions in other subjects:


Chemistry, 08.05.2021 14:00

Mathematics, 08.05.2021 14:00

World Languages, 08.05.2021 14:00

Health, 08.05.2021 14:00