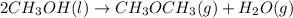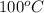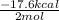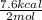Given the standard heat of combustion of methanol ch3oh is 182.6
kcal/mol, dimethyl ether ch3och3 is 347.6 kcal/mol, (methanol/
dimethyl ether in gas phase, water in liquid phase). given the heat
of vaporization of water is 10 kcal/mol, methanol is 8.4 kcal/mol,
dimethyl ether 4.8 kcal/mol. calculate the reaction of dehydration
of methanol to produce dimethyl ether. (indicate the phase of your
components).

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:00, dayjionneam
In the analysis of hair and fiber samples, which does a compound comparison microscope allow for that a conventional compound microscope does not? a. simultaneous observation b. polarization c. fluorescence d. higher magnification
Answers: 2

Chemistry, 21.06.2019 21:00, bakoeboo
Harvey kept a balloon with a volume of 348 milliliters at 25.0˚c inside a freezer for a night. when he took it out, its new volume was 322 milliliters, but its pressure was the same. if the final temperature of the balloon is the same as the freezer’s, what is the temperature of the freezer? the temperature of the freezer is kelvins.
Answers: 2


Chemistry, 22.06.2019 19:00, andrecoral105
A4.86 g piece of metal was placed in a graduated cylinder containing 15.5 ml of water. the water level rose to 17.3 ml. what is the density of the metal. i need the steps of how to solve it to so i can use a formula to work out other problems.
Answers: 1
You know the right answer?
Given the standard heat of combustion of methanol ch3oh is 182.6
kcal/mol, dimethyl ethe...
kcal/mol, dimethyl ethe...
Questions in other subjects:


Chemistry, 14.02.2020 00:31

Social Studies, 14.02.2020 00:31







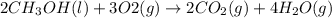 ....... (1)
....... (1)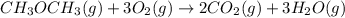 -347.6 kcal/mol
-347.6 kcal/mol
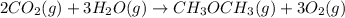 heat of reaction +347.6 kcal {sign reversed with reaction} ........ (2)
heat of reaction +347.6 kcal {sign reversed with reaction} ........ (2)