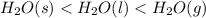Chemistry, 12.11.2019 21:31 msdmdsm1186
Place the following in order of increasing standard molar entropy h2o(l) h2o(g) h2o(s) a)h2o(s)< h2o(l)

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:30, ethanw8973
If 22.5 liters of oxygen reacted with excess of hydrogen, how many liters of water vapor could be produced?
Answers: 3

Chemistry, 22.06.2019 13:00, devontemiles8868
One of the hopes for solving the world's energy problem is to make use of the fusion reaction 21h +31h --> 42he + 10n + energy how much energy is released when 1 mol of deuterium is fused with 1 mol of tritium according to the above reaction? the masses of the atoms and the neutrons are as follows: 21h = 2.0140 amu 31h = 3.01605 amu 42he = 4.002603 amu 10n = 1.008665 amu. the speed of light is 2.9979 x 108 m/s.
Answers: 1

Chemistry, 22.06.2019 18:10, bri9263
Consider the following reaction at equilibrium: c(s)+h2o(g)⇌co(g)+h2(g) predict whether the reaction will shift left, shift right, or remain unchanged upon each of the following disturbances. a) c is added to the reaction mixture. b) h2ois condensed and removed from the reaction mixture c) co is added to the reaction mixture d) h2 is removed from the reaction mixture.
Answers: 3

Chemistry, 23.06.2019 01:30, bgarrison364
What is produced from neutralization of an acid and a base? a. hydronium ions b. citric acid c. salt and water
Answers: 1
You know the right answer?
Place the following in order of increasing standard molar entropy h2o(l) h2o(g) h2o(s) a)h2o(s)<...
Questions in other subjects:

History, 19.01.2021 17:00

English, 19.01.2021 17:00


Mathematics, 19.01.2021 17:00

History, 19.01.2021 17:00


English, 19.01.2021 17:00


Biology, 19.01.2021 17:00

Mathematics, 19.01.2021 17:00