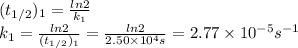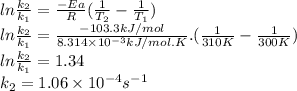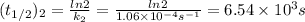For the reaction:
2n2o5(g) → 4no2(g) + o2(g) the rate law is: (δ[o2]/δt) = k[n2o5] at 300 k...

Chemistry, 08.11.2019 22:31 alexciamartinez05
For the reaction:
2n2o5(g) → 4no2(g) + o2(g) the rate law is: (δ[o2]/δt) = k[n2o5] at 300 k, the half-life is 2.50 × 104 seconds and the activation energy is 103.3 kj/mol. what is the half-life at 310 k? (hint: use rate law expression to determine the reaction order → solve for k1 at 300 k using the corresponding half-life expression → use two-point arrhenius equation to solve for k2 at 310 k → use the half-life expression again to solve for half-life at 310 k)

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:30, jabper5522
At a temperature of 393 k, the temperature of a sample of nitrogen is 1.07 atm what will the pressure be at a temperature of 478 k
Answers: 1


Chemistry, 22.06.2019 19:00, HaydenSturgis1
Which is the solubility product expression for caf2(s)?  [ca2+]/[f–]2  [ca2+][f2–]  [ca]+[f]2  [ca2+][f–]2
Answers: 3

Chemistry, 22.06.2019 21:50, isabel81ie
Given the data below for the reaction, 2 a + 2 b + 4 c => d + e + 3 f, the reaction is fill in the [ ] order in a, fill in the [ ] order in b, fill in the [ ] order in c and fill in the [ ] order overall. (use the words "first, second, third, fourth" to fill each blank)experimentinitial conc of a, mol/l initial conc of b, mol/l initial conc of c, mol/l initial rate, mol/l. s1 0.1 0.1 0.2 2 x 10-32 0.2 0.3 0.2 6 x 10-33 0.3 0.1 0.2 2 x 10-34 0.4 0.3 0.4 1.2 x 10-2
Answers: 2
You know the right answer?
Questions in other subjects:

Mathematics, 17.02.2021 16:20



English, 17.02.2021 16:20

Mathematics, 17.02.2021 16:20




Mathematics, 17.02.2021 16:20

Chemistry, 17.02.2021 16:30

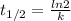
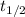 is the half-life
is the half-life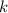 is the rate constant
is the rate constant