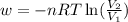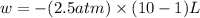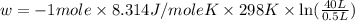Order the follow processes from (1) the least work done by the system to (5) the most work done by one mole of an ideal gas at 25°c. 1. an isothermal expansion from 1 l to 10 l at an external pressure of 2.5 atm. 2. a free isothermal expansion from 1 l to 100 l. 3. a reversible isothermal expansion from 0.5 l to 4 l. 4. a reversible isothermal expansion from 0.5 l to 40 l. 5. an isothermal expansion from 1 l to 100 l at an external pressure of 0.5 atm.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 21.06.2019 22:30, 20alondra04
In order to calculate the amount of heat transferred you must know the __ and specific heat of the material, as well as the change in temperature. a. volume b. density c. mass d. enthalpy
Answers: 1

Chemistry, 22.06.2019 07:20, mathman783
Why does his teacher ask him to balance the equation by including the correct coefficient
Answers: 1
You know the right answer?
Order the follow processes from (1) the least work done by the system to (5) the most work done by o...
Questions in other subjects:

Mathematics, 19.12.2019 20:31

Mathematics, 19.12.2019 20:31







History, 19.12.2019 20:31

Biology, 19.12.2019 20:31

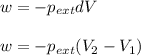
 = external pressure
= external pressure = initial volume of gas
= initial volume of gas = final volume of gas
= final volume of gas