
A0.05270.0527 g piece of metal containing an unknown amount of silver is dissolved to form 25.00 ml of solution. to the solution, 35.435.4 ml of 0.02190.0219 m kcl kcl is added, resulting in the complete precipitation of the silver ash agcl agcl . the excess cl−cl− added to the solution is then back titrated with a standard 0.02190.0219 m agno3 agno3 solution. a total of 14.3314.33 ml of the standard nano3 agno3 solution is required to reach the silver chromate end point in the mohr method. calculate the percent silver in the piece of metal.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:30, sammysosa121832
The ph of carrots are 5.0 how it is classified a. acidic b. basic c. indicator d. neutral
Answers: 2

Chemistry, 22.06.2019 08:30, itzhari101
In a chemical reaction at equilibrium, the rate of the forward reaction the rate of the reverse reaction. if the rate of the forward reaction more products are formed.
Answers: 1

Chemistry, 22.06.2019 13:00, monkeyrose1999
The molality of calcium chloride (cacl2) in an aqueous solution is 2.46 m. what is mole fraction of the solute?
Answers: 3
You know the right answer?
A0.05270.0527 g piece of metal containing an unknown amount of silver is dissolved to form 25.00 ml...
Questions in other subjects:

Health, 19.10.2020 23:01


History, 19.10.2020 23:01


History, 19.10.2020 23:01

Biology, 19.10.2020 23:01

Computers and Technology, 19.10.2020 23:01



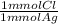 = 0.314 mmol Cl⁻
= 0.314 mmol Cl⁻


