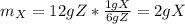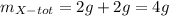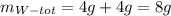It is required to make 12 grams of certain chemical compound called z. this is made from compounds w, x, and y in the ratio of 2: 1: 3. the compound y is itself made from w and x. to make 6 grams of y requires 4 grams of w and 2 grams of x. how much w and x is required to make the required amount of z.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 19:00, HaydenSturgis1
Which is the solubility product expression for caf2(s)?  [ca2+]/[f–]2  [ca2+][f2–]  [ca]+[f]2  [ca2+][f–]2
Answers: 3

Chemistry, 23.06.2019 00:30, zaniathomasel
What is bromine+calcium iodide--> calcium bromide +iodine balanced
Answers: 1

Chemistry, 23.06.2019 04:40, twinchristiansp4xhd2
6) (a) calculate the absorbance of the solution if its concentration is 0.0278 m and its molar extinction coefficient is 35.9 l/(mol cm). the depth of the cell is 5 mm. (b) what is the %t? (7) calculate the absorbance of the solution if the transmitted light intensity is 70% of the initial light beam intensity
Answers: 1
You know the right answer?
It is required to make 12 grams of certain chemical compound called z. this is made from compounds w...
Questions in other subjects:


Mathematics, 16.10.2020 07:01

Mathematics, 16.10.2020 07:01

Mathematics, 16.10.2020 07:01



Mathematics, 16.10.2020 07:01

Mathematics, 16.10.2020 07:01

Spanish, 16.10.2020 07:01