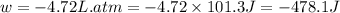Asample of gas in a cylinder of volume 3.42 l at 298 k and 2.57 atm expands to 7.39 l by two different pathways. path a is an isothermal, reversible expansion. path b has two steps. in the fi rst step, the gas is cooled at constant volume to 1.19 atm. in the second step, the gas is heated and allowed to expand against a constant external pressure of 1.19 atm until the fi nal volume is 7.39 l. calculate the work for each path.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 14:10, carsonjohnsonn
Can smoke be transformed into liquid or used as energy or both?
Answers: 2

Chemistry, 22.06.2019 12:30, quantamagic
Word equation for k(s)+h2o(l) yield koh(aq) + h2(g)
Answers: 1

Chemistry, 23.06.2019 05:00, contrerasdaisy100
Activity two: just lemons, inc. production here's a one-batch sample of just lemons lemonade production. determine the percent yield and amount of leftover ingredients for lemonade production and place your answers in the data chart. hint: complete stoichiometry calculations for each ingredient to determine the theoretical yield. complete a limiting reactant-to-excess reactant calculation for both excess ingredients. water sugar lemon juice lemonade percent yield leftover ingredients 946.36 g 196.86 g 193.37 g 2050.25 g 89% just lemons lemonade recipe equation: 2 water + sugar + lemon juice = 4 lemonade mole conversion factors: 1 mole of water = 1 cup = 236.59 g 1 mole of sugar = 1 cup = 225 g 1 mole of lemon juice = 1 cup = 257.83 g 1 mole of lemonade = 1 cup = 719.42 g show your calculations below. analysis questions 1. based on taste observations only, which ingredients were in excess in the lemonade samples in activity one? in activity one the excess substances for each sample were the water and sugar. 2. based on the data in activity two, which excess ingredients are affecting the taste of the lemonade in the sample batch? 3. what can just lemons, inc. do during production to reduce the amount of excess ingredients and improve the taste of their lemonade? 4. try to reduce the amount of leftover ingredients by changing the amount of one, two, or all three starting ingredients. show your stoichiometric calculations below. 5. during factory inspection, just lemons, inc. discovered that a water valve to the lemonade mixing station was not functioning. once they repair it, more water will enter the mixing station. from what you know about the limiting and excess ingredients for current lemonade production, what advice would you give engineers about the upcoming increase in water?
Answers: 3
You know the right answer?
Asample of gas in a cylinder of volume 3.42 l at 298 k and 2.57 atm expands to 7.39 l by two differe...
Questions in other subjects:


Social Studies, 23.07.2019 08:00

Mathematics, 23.07.2019 08:00

History, 23.07.2019 08:00






English, 23.07.2019 08:00

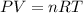
 = initial pressure of gas = 2.57 atm
= initial pressure of gas = 2.57 atm = initial volume of gas = 3.42 L
= initial volume of gas = 3.42 L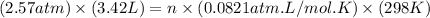

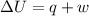
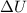 = internal energy
= internal energy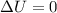


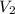 = final volume of gas = 7.39 L
= final volume of gas = 7.39 L
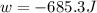

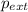 = external pressure = 1.19 atm
= external pressure = 1.19 atm