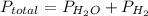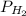Chemistry, 05.11.2019 02:31 jitosfc916
If the vapor pressure of water ( ) at 20.0°c is 17.5 mm hg, and the atmospheric pressure measured by a barometer (
) at 20.0°c is 17.5 mm hg, and the atmospheric pressure measured by a barometer ( ) was 757.3 mm hg, what is the partial pressure of h₂ gas (
) was 757.3 mm hg, what is the partial pressure of h₂ gas ( ) in the gas mixture in a buret? (use dalton’s law of partial pressures to solve for
) in the gas mixture in a buret? (use dalton’s law of partial pressures to solve for  )
)

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:30, jadepotts3965
Calculate the change in entropy if br2(l) is converted into gaseous br atoms. s° for br2(l) = 152.2 j/(mol•k) s° for br2(g) = 245.5 j/(mol•k) s° for br(g) = 175.0 j/(mol•k)
Answers: 2


Chemistry, 22.06.2019 05:00, foreignking02
1)each group 16 element has how many valence electrons? ( )4 ( )6 ( )8 ( )16 2)how many dots appear in the dot structure for calcium ion, ca2+? ( )zero ( )one ( )two ( )eight 3) which of the following atoms forms a cation to obtain an octet of outer shell electrons? ( )magnesium ( )oxygen ( )fluorine ( )helium 4) an al3+ ion contains 13 protons and 10 electrons. ( )true ( )false 5) valence and non-valence electrons are represented in lewis dot structures. ( )true ( )false
Answers: 3
You know the right answer?
If the vapor pressure of water ([tex]p_{h_2o}[/tex]) at 20.0°c is 17.5 mm hg, and the atmospheric pr...
Questions in other subjects:




Mathematics, 18.11.2019 04:31





Chemistry, 18.11.2019 04:31