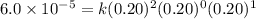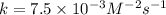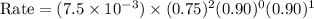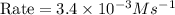Chemistry, 02.11.2019 03:31 aboatright7410
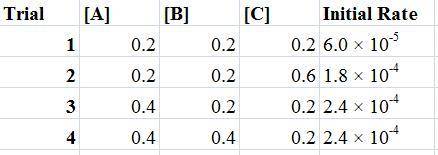
For the reaction a+b+c→d+e, the initial reaction rate was measured for various initial concentrations of reactants. the following data were collected: trial [a] (m) [b] (m) [c] (m) initial rate (m/s) 1 0.20 0.20 0.20 6.0×10−5 2 0.20 0.20 0.60 1.8×10−4 3 0.40 0.20 0.20 2.4×10−4 4 0.40 0.40 0.20 2.4×10−4for the reaction a+b+c -> d+e, the initial reaction rate was measured for various initial concentrations of reactants. the following data were collected: trial [a] (m) [b[ (m) [c] (m) initial rate (m/s)1 0.20 0.20 0.20 6.0 x 10^-52 0.20 0.20 0.60 1.8 x 10^-43 0.40 0.20 0.20 2.4 x 10^-44 0.40 0.40 0.20 2.4 x 10^-4reaction order respect to a = 2reaction order in respect to b = 0reaction order in respect to c = 1the value of the rate constant k for this reaction = 7.5*10^-3 m^-2 * s^-1given the data calculated in parts a, b, c, and d, determine the initial rate for a reaction that starts with 0.75m of reagent a and 0.90m of reagents b and c?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 14:10, gizmo50245
Calculate the mass percent of hydrogen in methyl acetate
Answers: 1



Chemistry, 22.06.2019 20:30, trevorhenyan51
What is a difference between a mixture of elements and a mixture of compounds
Answers: 1
You know the right answer?
For the reaction a+b+c→d+e, the initial reaction rate was measured for various initial concentration...
Questions in other subjects:

Mathematics, 12.12.2019 01:31

History, 12.12.2019 01:31

Mathematics, 12.12.2019 01:31



Mathematics, 12.12.2019 01:31


Mathematics, 12.12.2019 01:31

English, 12.12.2019 01:31

Social Studies, 12.12.2019 01:31

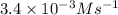
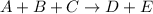
![\text{Rate}=k[A]^a[B]^b[C]^c](/tpl/images/0356/3589/be89a.png)
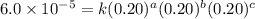 ....(1)
....(1)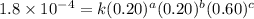 ....(2)
....(2)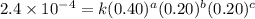 ....(3)
....(3)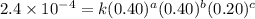 ....(4)
....(4)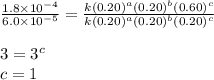
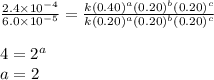
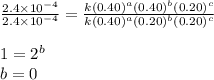
![\text{Rate}=k[A]^2[B]^0[C]^1](/tpl/images/0356/3589/54afd.png)