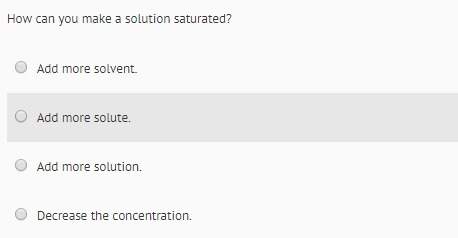
The bond strength of compounds a, b, c, and d as measured by their bond energies
(kj/mol 350,...

Chemistry, 01.11.2019 19:31 theoriginalstal9245
The bond strength of compounds a, b, c, and d as measured by their bond energies
(kj/mol 350, 500, 180, and 1,450, respectively. which compound will most likely
conduct electricity when dissolved in water?
o compound a
compound b
o compound c
compound d

Answers: 1


Other questions on the subject: Chemistry




Chemistry, 22.06.2019 08:00, wizz4865
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2
You know the right answer?
Questions in other subjects:



Social Studies, 17.07.2019 23:00







Social Studies, 17.07.2019 23:00