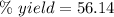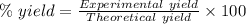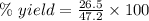Chemistry, 01.11.2019 03:31 tamikagoss22
The theoretical yield of a reaction can be determined using its in the reaction between co and fe3o4, the theoretical yield in an experiment is calculated to be 47.2 g fe. when a careless chemistry student carries out the experiment, the actual yield is 26.5 g fe. calculate the percentage yield. chemical equation and the starting amounts of the reactants.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:00, foreignking02
1)each group 16 element has how many valence electrons? ( )4 ( )6 ( )8 ( )16 2)how many dots appear in the dot structure for calcium ion, ca2+? ( )zero ( )one ( )two ( )eight 3) which of the following atoms forms a cation to obtain an octet of outer shell electrons? ( )magnesium ( )oxygen ( )fluorine ( )helium 4) an al3+ ion contains 13 protons and 10 electrons. ( )true ( )false 5) valence and non-valence electrons are represented in lewis dot structures. ( )true ( )false
Answers: 3


You know the right answer?
The theoretical yield of a reaction can be determined using its in the reaction between co and fe3o4...
Questions in other subjects:

Chemistry, 03.12.2021 15:30

Mathematics, 03.12.2021 15:40

English, 03.12.2021 15:40

Mathematics, 03.12.2021 15:40


Mathematics, 03.12.2021 15:40

Mathematics, 03.12.2021 15:40


Biology, 03.12.2021 15:40