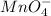Chemistry, 30.10.2019 05:31 amandasantiago2001
(2 points) dumbledore thinks the students are now confident with significant figures; snape is not so confident of begrudgingly snape decides to move forward in his lesson. his next lesson is in naming ionic compounds. he presents his students with sodium hydrogen sulfate and potassium permanganate. which pair of ionic formulas is correct for these two compounds?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:00, tbiles99
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1

Chemistry, 22.06.2019 14:10, roserose3098
16. in a reaction that has reached equilibrium, a. the forward and reverse reactions are occurring at the same rate. b. the reactants and products are in equal concentrations. c. the forward reaction has gone further than the reverse reaction. d. there are equal numbers of atoms on both sides of the equation. e. a, b, and d are correct.
Answers: 2

Chemistry, 22.06.2019 22:00, notearslefttocry14
Imagine one batch of soup (batch “a”) is made with 8.19 g/can of salt, according to the recipe, and a second batch of soup (batch “b”) is made with 8.32 g/can of salt. explain which batch would be more resistant to frost damage if it is shipped a great distance in winter and explain why.
Answers: 2

Chemistry, 23.06.2019 01:30, emfranco1
Which of the following statements is true about energy quantization at the atomic level? electrons in the outermost orbits are the most stable. electrons in all the orbits around the nucleus have the same amount of energy. electrons in the orbit closest to the nucleus have the least amount of energy. electrons absorb or release the same amount of energy independent of the energy levels.
Answers: 1
You know the right answer?
(2 points) dumbledore thinks the students are now confident with significant figures; snape is not...
Questions in other subjects:




Mathematics, 19.11.2020 20:10


Biology, 19.11.2020 20:10



Mathematics, 19.11.2020 20:10

Mathematics, 19.11.2020 20:10

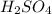 and
and 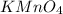 respectively.
respectively. .
.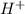 ion, this element will loose 1 electron.
ion, this element will loose 1 electron.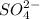
![[Ar]4s^1](/tpl/images/0352/3370/85db5.png) .
.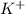 ion, this element will loose 1 electron.
ion, this element will loose 1 electron.