Chemistry, 25.10.2019 19:43 niceguy1997
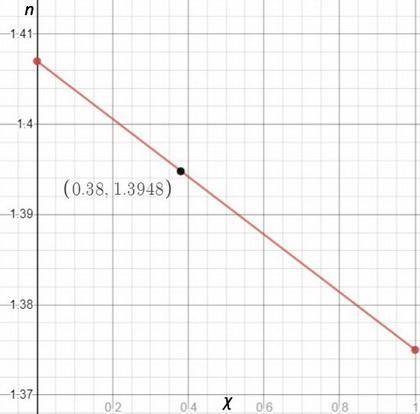
You are performing a simple distillation of roughly 50: 50 liquid solution containing two components, hexane and nonane you place 15 ml of the mixture in a round bottom flask, and then collect the distillate sequentially as four three ml samples labeled s1, s2, s3, and s4. pure hexane has a refractive index of 1.375 and pure nonane has a refractive index of 1.407. you measure a refractive index of 1.3948 for one of the four samples. assuming the refractive index varies linearly with mole fraction, estimate the mole fraction of hexane in this sample.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 04:50, shonnybenskin8
Compare the equilibrium constants for the systems shown in the table. which favors products the most? which favors products the least? rank these systems in order from most to least in terms of favoring products rather than reactants. d > b > a > c c > a > b > d b > c > d > a a > d > c > b
Answers: 1

Chemistry, 22.06.2019 06:00, tddreviews
Match the name of the following compound: mgso4 · h2omagnesium sulfate monohydratemagnesium (ii) sulfate monohydratemagnesium (ii) sulfate hydratemagnesium sulfate hydrate
Answers: 1

Chemistry, 22.06.2019 08:30, Blaise2653
Joan writes four numbers on the board in standard form, and then she writes their scientific notation
Answers: 1
You know the right answer?
You are performing a simple distillation of roughly 50: 50 liquid solution containing two components...
Questions in other subjects:


Mathematics, 04.12.2020 18:00




English, 04.12.2020 18:00

Chemistry, 04.12.2020 18:00