
Chemistry, 31.01.2020 16:52 hardwick744
Calculate the ph of an aqueous solution of 16 ml of 0.00046 m hclo4(aq) after dilution to 42 ml.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 13:20, monsurviky
Amixture of gaseous sulfur dioxide and oxygen are added to a reaction vessel and heated to 1000 k where they react to form so3(g). if the vessel contains 0.669 atm so2(g), 0.395 atm o2(g), and 0.0851 atm so3(g) after the system has reached equilibrium, what is the equilibrium constant kp for the reaction: 2 so2(g) o2(g) ⇌ 2 so3(g)
Answers: 3


Chemistry, 22.06.2019 02:30, drivinghydra
What is the relation between concentration of reactants and the rate of chemical reaction?
Answers: 1

You know the right answer?
Calculate the ph of an aqueous solution of 16 ml of 0.00046 m hclo4(aq) after dilution to 42 ml....
Questions in other subjects:


English, 30.03.2021 23:40

Mathematics, 30.03.2021 23:40

Mathematics, 30.03.2021 23:40





Mathematics, 30.03.2021 23:40

Mathematics, 30.03.2021 23:40

 of HClO_{4} [/tex] contain 0.00046mol
of HClO_{4} [/tex] contain 0.00046mol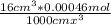
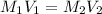
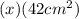

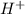 ]
]


