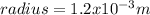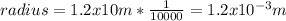Chemistry, 23.10.2019 02:00 ashioya0021
The radius of an atom of element x is 1.2x10m the radius of the centre of the atom is 1/1 the radius of the atom calculate the radius of the centre of an atom of element x

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:00, hemolelekeakua
The alkali metals (group 1) consist of lithium (3), sodium (11), potassium (19), rubidium (37), cesium (55), and francium (87). they are soft, metallic solids with low densities and low melting points. based on the data shown in figure 1, how many valence electrons do alkali metals share?
Answers: 3


Chemistry, 22.06.2019 12:30, hayleyconsole
Nebulae are enormous clouds in outer space. they are made mostly of hydrogen gas, helium gas, and dust. some nebulae glow brightly, while others do not. the stars that people see are huge, bright balls of glowing gas. they are made mostly of hydrogen and helium. which statement correctly describes other ways in which nebulae and stars are different? a. stars can form inside a nebula but a nebula can never be produced by any star. b. a star always has a higher density than a nebula. c. stars can never form inside a nebula but a nebula can be produced by any star. d. a nebula always has a higher density than a star.
Answers: 3
You know the right answer?
The radius of an atom of element x is 1.2x10m the radius of the centre of the atom is 1/1 the radius...
Questions in other subjects:

Health, 15.12.2020 01:40

Mathematics, 15.12.2020 01:40


Chemistry, 15.12.2020 01:40


Mathematics, 15.12.2020 01:40

Mathematics, 15.12.2020 01:40


History, 15.12.2020 01:40

Mathematics, 15.12.2020 01:40