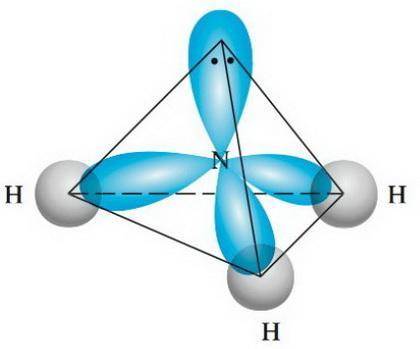
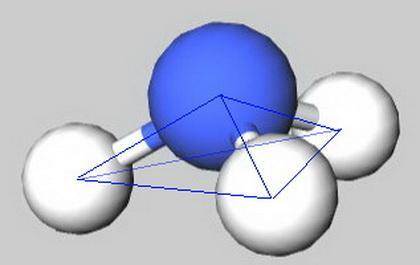
Considering the definition of electron geometry and molecule geometry, the main difference between electronic geometry and molecular geometry is that electronic geometry includes the bonds and pairs of electrons in the molecule, whereas molecular geometry includes only the number of bonds that are present in a molecule.
Molecular geometry
Molecular geometry refers to the three-dimensional arrangement of the atoms that make up a molecule. It is a determinant of many of the compound's properties, for example, boiling point, density, or solubility.
Molecular geometry can be predicted using the Valencia Shell Electronic Pair Repulsion Theory, which is based on the fact that electrons tend to repel each other (due to similar charges). For this reason, the orbitals that contain the electrons are oriented in such a way that they are as far away from each other.
Electronic geometry
On the other hand, electronic geometry is the way in which electrons are arranged in space, considering all the valence pairs of the central atom. Generally matches molecular geometry but not always.
Difference
So, the electron geometry depends on the structure of the electrons of the central atom of a molecule, while the molecular geometry depends on whether other atoms are attached to the central atom or if there are pairs of free electrons.
Learn more:
linklink