
Chemistry, 18.10.2019 03:30 tdyson3p6xvtu
2. suppose that 21.37 ml of naoh is needed to titrate 10.00 ml of 0.1450 m h2so4 solution.
(a) write a balanced reaction equation for the acid-base neutralization.
(b) calculate the molarity of naoh based on the results of this titration.

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 06:00, palomaresmitchelle
There are 6.022, 104 atoms of hg in 1 mole of hg the number of atoms in 45 moles of hg can be found by multiplying 4.5 by 6.022, 102 which is the number of atoms in 4.5 moles of hg, correctly written in scientific notation with the correct number of significant figures? 0 21,109 0 21,100 271, 1024 27.099, 100 mark this and retum save and exit submit
Answers: 1

Chemistry, 22.06.2019 09:40, kolibeilfuss
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 10:00, 2019reynolds
Which sentence about particles in matter is true? a. atoms are present in solids and liquids but not in gases. b. the particles of matter are in constant motion. c. the same kinds of atoms are found in different elements. d. when a solid changes to a liquid, the sizes of the particles change.
Answers: 1
You know the right answer?
2. suppose that 21.37 ml of naoh is needed to titrate 10.00 ml of 0.1450 m h2so4 solution.
(a)...
(a)...
Questions in other subjects:


History, 26.07.2019 05:00



Mathematics, 26.07.2019 05:00




Mathematics, 26.07.2019 05:00

Health, 26.07.2019 05:00

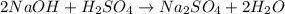
 can be calculated as:
can be calculated as:




