
Chemistry, 17.10.2019 18:30 lalalaaldgydyhg8823
Determine the [oh−] , ph, and poh of a solution with a [h+] of 2.2×10−12 m at 25 °c. [oh−]= m ph= poh= determine the [h+] , ph, and poh of a solution with an [oh−] of 9.7×10−5 m at 25 °c. [h+]= m ph= poh= determine the [h+] , [oh−] , and poh of a solution with a ph of 10.30 at 25 °c. [h+]= m [oh−]= m poh= determine the [h+] , [oh−] , and ph of a solution with a poh of 9.06 at 25 °c. [h+]= m [oh−]= m ph=

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, erikloza12pdidtx
Which type of bond is present in hydrogen sulfide (h2s)? the table of electronegativities is given. a. hydrogen b. ionic c. nonpolar covalent d. polar covalent
Answers: 1

Chemistry, 22.06.2019 02:30, dijonmckenzie3
Margaret wants to make an orange flavored drink by stirring powdered drink mix into a glass of water. she doesn't like drinks that have small clumps of powdered solid in them, so she wants the drink to be a perfect solution. what factors should margaret not consider when deciding how much powder to add to her glass of water?
Answers: 3

Chemistry, 22.06.2019 05:50, vanessa051266
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1
You know the right answer?
Determine the [oh−] , ph, and poh of a solution with a [h+] of 2.2×10−12 m at 25 °c. [oh−]= m ph= po...
Questions in other subjects:

Mathematics, 30.10.2019 05:31


Mathematics, 30.10.2019 05:31


History, 30.10.2019 05:31


History, 30.10.2019 05:31

Mathematics, 30.10.2019 05:31

English, 30.10.2019 05:31

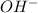 , pH and pOH of the solution is
, pH and pOH of the solution is 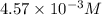 , 11.66 and 2.36 respectively.
, 11.66 and 2.36 respectively.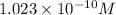 , 4.01 and 9.99 respectively.
, 4.01 and 9.99 respectively.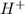 ,
, 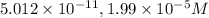 and 3.7 respectively.
and 3.7 respectively.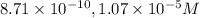 and 4.97 respectively.
and 4.97 respectively.![pH=-\log[H^+]](/tpl/images/0329/0431/cf945.png) .....(1)
.....(1)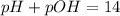 .....(2)
.....(2)![[OH^]](/tpl/images/0329/0431/ffd27.png) of the solution, we use the equation:
of the solution, we use the equation:![pOH=-\log[OH^-]](/tpl/images/0329/0431/fe336.png) ......(3)
......(3)![[H^+]=2.2\times 10^{-12}M](/tpl/images/0329/0431/bd40e.png)
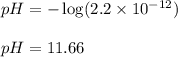
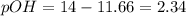
![2.36=-\log[OH^-]](/tpl/images/0329/0431/c7189.png)
![[OH^-]=10^{-2.36}=4.57\times 10^{-5}M](/tpl/images/0329/0431/7f1bf.png)
![[H^+]=9.7\times 10^{-5}M](/tpl/images/0329/0431/f8e3a.png)
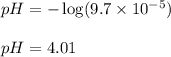
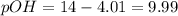
![9.99=-\log[OH^-]](/tpl/images/0329/0431/2903e.png)
![[OH^-]=10^{-9.99}=1.023\times 10^{-10}M](/tpl/images/0329/0431/9422a.png)
![10.30=-\log[H^+]](/tpl/images/0329/0431/05afc.png)
![[H^+]=10^{-10.30)=5.012\times 10^{-11}M](/tpl/images/0329/0431/9890d.png)
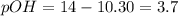
![3.7=-\log[OH^-]](/tpl/images/0329/0431/1954f.png)
![[OH^-]=10^{-3.7}=1.99\times 10^{-4}M](/tpl/images/0329/0431/a2180.png)
![9.03=-\log[H^+]](/tpl/images/0329/0431/15752.png)
![[H^+]=10^{-9.30)=8.71\times 10^{-10}M](/tpl/images/0329/0431/070a8.png)
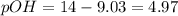
![4.97=-\log[OH^-]](/tpl/images/0329/0431/c7d38.png)
![[OH^-]=10^{-4.97}=1.07\times 10^{-5}M](/tpl/images/0329/0431/10104.png)



