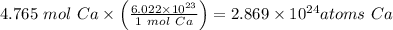Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:30, ciarakelly636owuiup
Asample of the male sex hormone testosterone, c19h28o2, contains 3.88×10^21 atoms of hydrogen.(a) how many atoms of carbon does it contain? (b) how many molecules of testosterone does it contain? (c) how many moles of testosterone does it contain? (d) what is the mass of this sample in grams?
Answers: 1


Chemistry, 22.06.2019 05:10, hadellolo8839
How many miles of water are produced if 5.43 mol pbo2 are consumed
Answers: 1

You know the right answer?
How many atoms are in 191 g of calcium...
Questions in other subjects:



Mathematics, 24.03.2021 18:30

Mathematics, 24.03.2021 18:30

English, 24.03.2021 18:30

Social Studies, 24.03.2021 18:30


Mathematics, 24.03.2021 18:30

Social Studies, 24.03.2021 18:30

Mathematics, 24.03.2021 18:30

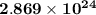 atoms Ca
atoms Ca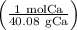 =4.765 mol Ca
=4.765 mol Ca particles mol, calculate the number of atoms present
particles mol, calculate the number of atoms present