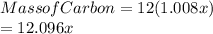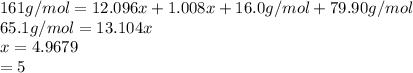Chemistry, 12.10.2019 06:10 whitakers87
Acompound of molar mass 161 g/mol contains only carbon, hydrogen, bromine, and oxygen. analysis reveals that the compound contains 12 times as much carbon as hydrogen by mass. find the molecular formula.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:00, tamikagoss22
Agas in a balloon at constant pressure has a volume of 160 ml at -125*c. what is its volume at 29.0*c?
Answers: 1

Chemistry, 21.06.2019 22:00, breannaasmith1122
If 1.63 times 10 negative 4 of helium dissolves in 100.0g of water, what is the concentration in parts per million
Answers: 3

Chemistry, 22.06.2019 07:30, gwenparks
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1

Chemistry, 22.06.2019 10:30, esnyderquintero
How do you lengthen a pattern piece? (family and consumer science, sewing)
Answers: 2
You know the right answer?
Acompound of molar mass 161 g/mol contains only carbon, hydrogen, bromine, and oxygen. analysis reve...
Questions in other subjects:


Arts, 29.10.2019 15:31


Chemistry, 29.10.2019 15:31

History, 29.10.2019 15:31

Biology, 29.10.2019 15:31

Mathematics, 29.10.2019 15:31