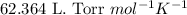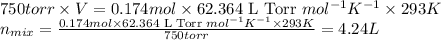Some commercial drain cleaners contain two components: sodium hydroxide and aluminum in the form of a powder. when the mixture is poured down a clogged drain, the following redox reactions occurs: 2naoh(aq) + 2 al(s) + 6h2o(l) → 2naal(oh)4(aq) + 3 h2(g) the heat generated in this reaction melt away grese and the hydrogen gas released stirs up the solids clagging the drain. calculate the volume hydrogen gas formed at 20. ºc and 750. torr if 3.12 g of al is treated with excess naoh.

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 09:20, payshencec21
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1


Chemistry, 22.06.2019 12:30, nekathadon
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1
You know the right answer?
Some commercial drain cleaners contain two components: sodium hydroxide and aluminum in the form of...
Questions in other subjects:


Chemistry, 07.12.2020 17:00


Mathematics, 07.12.2020 17:00


English, 07.12.2020 17:00




Mathematics, 07.12.2020 17:00

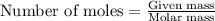
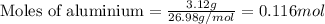
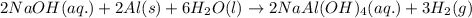
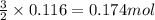 of hydrogen gas
of hydrogen gas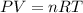
![20^oC=[20+273]K=293K](/tpl/images/0308/3868/3b5d4.png)