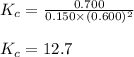At equilibrium, the concentrations of reactants and products can be predicted using the equilibrium constant, kc, which is a mathematical expression based on the chemical equation. for example, in the reaction aa+bb⇌cc+dd where a, b, c, and d are the stoichiometric coefficients, the equilibrium constant is kc=[c]c[d]d[a]a[b]b where [a], [b], [c], and [d] are the equilibrium concentrations. if the reaction is not at equilibrium, the quantity can still be calculated, but it is called the reaction quotient, qc, instead of the equilibrium constant, kc. qc=[c]tc[d]td[a]ta[b]tb where each concentration is measured at some arbitrary time t. part a a mixture initially contains a, b, and c in the following concentrations: [a] = 0.350 m , [b] = 0.800 m , and [c] = 0.500 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.150 m and [c] = 0.700 m . calculate the value of the equilibrium constant, kc. express your answer numerically.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:00, maddynichole2017
Read these sentences from the text. near the equator, the tropics receive the most rain on a consistent basis. as a result, the fresh water falling into the ocean decrease the salinity of the surface water in that region. [. .] . . as the salt content of sea water increases, so does its density. what can you infer about how rain affects the density of surface water near the equator?
Answers: 1


Chemistry, 22.06.2019 09:50, Amandachavez94
Although respiratory organs vary across different organisms, they all contain respiratory surfaces that have a large surface area and are extremely thin. explain why having an extremely thin respiratory surface with a large surface area is advantageous for the process of gas exchange
Answers: 1
You know the right answer?
At equilibrium, the concentrations of reactants and products can be predicted using the equilibrium...
Questions in other subjects:

Spanish, 13.03.2021 08:20

English, 13.03.2021 08:20

Mathematics, 13.03.2021 08:20


Mathematics, 13.03.2021 08:20

English, 13.03.2021 08:20



Mathematics, 13.03.2021 08:20

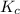
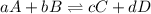
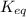 is written as:
is written as:![K_{eq}=\frac{[C]^c[D]^d}{[A]^a[B]^b}](/tpl/images/0304/8312/9c8b0.png)
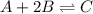
![[A]_{eq}=0.150M](/tpl/images/0304/8312/2394d.png)
![[C]_{eq}=0.700M](/tpl/images/0304/8312/9e4dd.png)
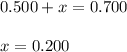
![[B]_{eq}=(0.800-x)=0.800-0.200=0.600M](/tpl/images/0304/8312/eb193.png)
![K_c=\frac{[C]}{[A][B]^2}](/tpl/images/0304/8312/240ef.png)