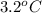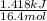Acommon laboratory reaction is the neutralization of an acid with a base. when 32.8 ml of 0.500 m hcl at 25.0°c is added to 66.3 ml of 0.500 m naoh at 25.0°c in a coffee cup calorimeter (with a negligible heat capacity), the temperature of the mixture rises to 28.2°c. what is the heat of reaction per mole of nacl (in kj/mol)? assume the mixture has a specific heat capacity of 4.18 j/(g·k) and that the densities of the reactant solutions are both 1.07 g/ml.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 19:50, strawberrymrmr756
Which sentence from holes contains an implied personality trait? stanley and his parents had tried to pretend that he was just going away to camp for a while, just like rich kids do. he'd just been in the wrong place at the wrong time. stanley felt somewhat dazed as the guard unlocked his handcuffs and led him off the bus. stanley nodded to show he understood
Answers: 3

Chemistry, 22.06.2019 20:00, bbyjean9974
State one important difference between a physical change and a chemical change?
Answers: 1


Chemistry, 22.06.2019 22:30, brianna5626
How do limiting factors most affect population size? ostop population growthrestrict population growthincrease population sizeresult in positive impactso
Answers: 1
You know the right answer?
Acommon laboratory reaction is the neutralization of an acid with a base. when 32.8 ml of 0.500 m hc...
Questions in other subjects:


Mathematics, 21.04.2021 18:10

Computers and Technology, 21.04.2021 18:10

Mathematics, 21.04.2021 18:10


SAT, 21.04.2021 18:10



History, 21.04.2021 18:10

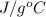



 =
=