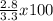Chemistry, 08.10.2019 01:00 algahimnada
Calcium oxide reacts with water in a combination reaction to produce calcium hydroxide: cao(s) h2o(l) → ca(oh)2(s) in a particular experiment, a 2.50-g sample of cao is reacted with excess water and 2.80 g of ca(oh)2 is recovered. what is the percent yield in this experiment

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, pinkycupcakes3oxbqhx
200. ml of 3.00 m nacl solution is diluted to a final volume of 500. ml. what is the molarity of the final solution?
Answers: 2

Chemistry, 22.06.2019 17:00, brownvester44
Astable electron arrangement for an atom is one that does not easily change. how is this arrangement arrived at? a. valence electrons are transferred or shared to create a full outer shell of electrons. b. valence electrons are discarded into space to create a full outer shell of electrons. c. protons (positive charge) pair with valence electrons (negative charge) to create a strong bond. d. outer shells with valence electrons are transferred or shared.
Answers: 2

Chemistry, 22.06.2019 17:10, mikeeway33
Some liquids can be distilled, but only at temperatures that are so high that it is impractical, or so high the compound decomposes. explain why distillation such compounds at significantly less than atmospheric pressure (some degree of vacuum) would solve this problem.
Answers: 2
You know the right answer?
Calcium oxide reacts with water in a combination reaction to produce calcium hydroxide: cao(s) h2o(...
Questions in other subjects:



Business, 26.07.2019 23:30

Biology, 26.07.2019 23:30


History, 26.07.2019 23:30


Mathematics, 26.07.2019 23:30

Biology, 26.07.2019 23:30

Mathematics, 26.07.2019 23:30