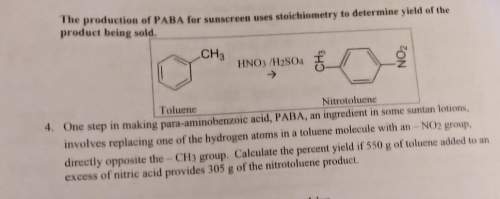
One step in making para-aminobenzoic acid, paba, an ingredient in some suntan lotions,
involves...

Chemistry, 06.10.2019 22:00 IdkHowToDoMath
One step in making para-aminobenzoic acid, paba, an ingredient in some suntan lotions,
involves replacing one of the hydrogen atoms in a toluene molecule with an - no2 group,
directly opposite the -ch3 group. calculate the percent yield if 550 g of toluene added to an
excess of nitric acid provides 305 g of the nitrotoluene product.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:30, salvadorperez26
Suppose that during that icy hot lab 65,000 j of energy were transferred to 450 g of water at 20°c what would have have been the final temperature of the water
Answers: 2


Chemistry, 22.06.2019 13:30, richardwalker8ourhg2
An animal cell loses the ability to convert energy stored in food to energy that the cell can use. which of the cell's organelles has stopped working? a. the mitochondria b. the nucleus c. the vacuoles d. the endoplasmic reticulum
Answers: 1

Chemistry, 22.06.2019 14:30, joejoefofana
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀ pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4. 0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3
You know the right answer?
Questions in other subjects:





Mathematics, 22.02.2021 21:00

Mathematics, 22.02.2021 21:00


Mathematics, 22.02.2021 21:00

Mathematics, 22.02.2021 21:00



