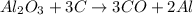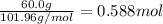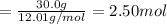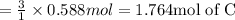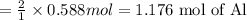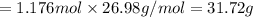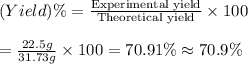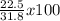Chemistry, 06.10.2019 04:20 andrewmena05
The reaction of 60.0 g of aluminum oxide with 30.0 g of carbon produced co gas and 22.5 g of aluminum metal. what is the percent yield aluminum for this reaction?

Answers: 1


Other questions on the subject: Chemistry




Chemistry, 22.06.2019 20:20, carcon2019
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3
You know the right answer?
The reaction of 60.0 g of aluminum oxide with 30.0 g of carbon produced co gas and 22.5 g of aluminu...
Questions in other subjects:

Mathematics, 12.07.2021 19:30



Mathematics, 12.07.2021 19:30