
Chemistry, 02.10.2019 22:00 TropicalFan
Certain atoms emit photons of light with an energy of 3.820 ✕ 10−19 j. calculate the frequency (in hz) and wavelength (in nm) of one of these photons.
frequency hz
and
wavelength nm
what is the total energy (in kj) in 1 mole of these photons?
kj

Answers: 2


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 11:00, blondieb1722
Which are examples of how technology has advanced scientific understanding.1using hot water to sterilize medical equipment.2transplanting a human organ into another individual.3inserting genes from one sheep into another cell to make a cloneunderstanding the different structures that make up a cell.4examining microorganisms from the deepest parts of the ocean
Answers: 2
You know the right answer?
Certain atoms emit photons of light with an energy of 3.820 ✕ 10−19 j. calculate the frequency (in h...
Questions in other subjects:



Social Studies, 12.02.2021 07:20

Mathematics, 12.02.2021 07:20

Mathematics, 12.02.2021 07:20

Chemistry, 12.02.2021 07:20

Social Studies, 12.02.2021 07:20


Spanish, 12.02.2021 07:20

Mathematics, 12.02.2021 07:20

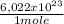 = 230040 J/mole ≡ 230,0 kJ/mole
= 230040 J/mole ≡ 230,0 kJ/mole

