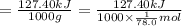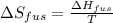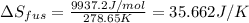Chemistry, 02.10.2019 21:10 CameronVand21
The enthalpy of fusion for benzene (c6h6, 78.0 g/mol) is 127.40 kj/kg, and its melting point is 5.5°c. what is the entropy change when 1 mole of benzene melts at 5.5°c? (show all unit conversions and write out all equations)

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, nmadrigal
In a laboratory experiment, a fermenting aqueous solution of glucose and yeast produces carbon dioxide gas and ethanol. the solution was heated by burning natural gas in a bunsen burner to distill the ethanol that formed in the flask. during the distillation, the ethanol evaporated and then condensed in the receiving flask. the flame of the burner was kept too close to the bottom of the flask and some of the glucose decomposed into a black carbon deposit on the inside of the flask. during this experiment the following changes occurred. which of these changes involved a physical change and not a chemical change? check all that apply. 1-condensation of ethanol 2-evaporation of ethanol 3- formation of carbon dioxide gas from glucose burning of natural gas 4-formation of ethanol from glucose by yeast 5-formation of a carbon deposit inside the flask
Answers: 2

Chemistry, 22.06.2019 05:30, greekfreekisdbz
What type of reaction is shown below? check all that apply. 2h2o2 → 2h2o + o2 synthesis decomposition combustion
Answers: 3

Chemistry, 23.06.2019 02:30, hailee232
When the ionic compound nabr dissolves in water, br– ions are pulled into solution by the attraction between what two particles? a. the na+ and br– ions b. the na+ ion and the negative end of a water molecule c. the br– ion and the positive end of a water molecule d. the br– ion and the negative end of a water molecule
Answers: 1

Chemistry, 23.06.2019 04:10, nabeelunique
An unknown substance has been shown to have metallic bonds. which of the following is most likely a property of this substance? a. low conductivity b. low boiling point c. high malleability d. high solubility in water
Answers: 2
You know the right answer?
The enthalpy of fusion for benzene (c6h6, 78.0 g/mol) is 127.40 kj/kg, and its melting point is 5.5°...
Questions in other subjects:






Mathematics, 15.04.2020 20:49


Spanish, 15.04.2020 20:49


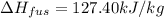
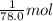
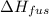 = 127.40 kJ/kg=127.40 kJ/1000 g (1kg = 1000 g)
= 127.40 kJ/kg=127.40 kJ/1000 g (1kg = 1000 g)