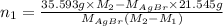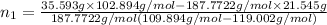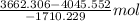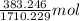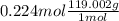Chemistry, 01.10.2019 22:00 GodlyGamer8239
Both kbr and nabr are soluble ionic compounds and fully dissociate in aqueous solution. a 21.545-g mixture of kbr and nabr is dissolved in water, then a solution of agno3 is added so that all of the bromine present is converted to solid agbr. the agbr product is dried and found to have a mass of 35.593 g. what mass of kbr was present in the original mixture?

Answers: 1


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 11:00, 21villalobosjabez
Which type of fossil does this image depict?
Answers: 1
You know the right answer?
Both kbr and nabr are soluble ionic compounds and fully dissociate in aqueous solution. a 21.545-g m...
Questions in other subjects:



English, 01.07.2019 01:30


History, 01.07.2019 01:30



Biology, 01.07.2019 01:30


Computers and Technology, 01.07.2019 01:30

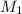 and NaBr is
and NaBr is 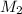 .
.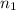 and
and 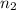 .
.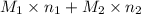 = 21.545 g
= 21.545 g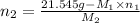
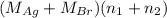 = 35.593 g
= 35.593 g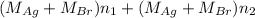 = 35.593 g
= 35.593 g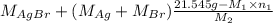 = 35.593 g
= 35.593 g