
Astudent determines the molar mass of a compound by the method used in this experiment. she found the temperature of the ice water to be 1.0°c. when she added 11.1g of her unknown solute to the mixture, the temperature fell to -3.0°c. the mass of the solution was 90.4g. calculate mass of solute in solution 1kg of water

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:00, parisaidan366
What happened in 2012 and how does it illustrate the importance of understanding the sun and how it works?
Answers: 3


Chemistry, 22.06.2019 09:20, pandaman632
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1

Chemistry, 22.06.2019 09:40, kolibeilfuss
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3
You know the right answer?
Astudent determines the molar mass of a compound by the method used in this experiment. she found th...
Questions in other subjects:

Biology, 10.07.2019 03:30

History, 10.07.2019 03:30

History, 10.07.2019 03:30

History, 10.07.2019 03:30

History, 10.07.2019 03:30

History, 10.07.2019 03:30

History, 10.07.2019 03:30

History, 10.07.2019 03:30

History, 10.07.2019 03:30

History, 10.07.2019 03:30

 =-3.0°C
=-3.0°C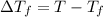
 =1.0°C-( -3.0°C)=4°C
=1.0°C-( -3.0°C)=4°C
 = Molal depression constant
= Molal depression constant


 g of solute /kg of water
g of solute /kg of water

