
Chemistry, 28.09.2019 02:30 ayoismeisjuam
Consider the gas reaction: h2 (g)2(g) 2hi (g) the equilibrium constant at 731 k is 50.3. equal amounts of all three gases (0.100 m) are introduced in a container, calculate the concentration of each gas after the system reaches equilibrium. express your results with the right number of significant figures.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:00, Britny2386
2h2s + 3o2 2so2 + 2h2o which option gives the correct mole ratios? h2s: so2 = 2: 2 and o2: h2o = 3: 2 h2s: so2 = 2: 3 and o2: h2o = 3: 2 h2s: so2 = 4: 4 and o2: h2o = 5: 4 h2s: so2 = 4: 6 and o2: h2o = 4: 4
Answers: 1


Chemistry, 22.06.2019 10:30, kluckey3426
Asample of air with a volume of 2.20m3 at a pressure of 105 kpa and a temperature of 30c is cooled to 10c and the pressure is reduced to 75.0 kpa. what is the new volume? 6.9 1.34 2.56 43.0 2.88
Answers: 1

Chemistry, 22.06.2019 12:00, Unknowndragon42
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2
You know the right answer?
Consider the gas reaction: h2 (g)2(g) 2hi (g) the equilibrium constant at 731 k is 50.3. equal amou...
Questions in other subjects:

Mathematics, 06.05.2020 06:28

Arts, 06.05.2020 06:28


Mathematics, 06.05.2020 06:28

Mathematics, 06.05.2020 06:28



Mathematics, 06.05.2020 06:28


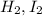 and
and 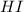 at equilibrium 0.033 M, 0.033 M and 0.234 M respectively.
at equilibrium 0.033 M, 0.033 M and 0.234 M respectively.

 will be,
will be,![K=\frac{[HI]^2}{[H_2][I_2]}](/tpl/images/0269/8551/8a740.png)


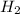 and
and 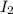 at equilibrium = (0.100-x) = 0.100 - 0.067 = 0.033 M
at equilibrium = (0.100-x) = 0.100 - 0.067 = 0.033 M

