Chemistry, 28.09.2019 01:30 daltonrebekah3532
Propane can be turned into hydrogen by the two-step reforming process. in the first step, propane and water react to form carbon monoxide and hydrogen: c3hg(g)+3 3 co(g)+7 h2(g) in the second step, carbon monoxide and water react to form hydrogen and carbon dioxide: co(g)+h20(g)h2(9)+co2(g) write the net chemical equation for the production of hydrogen from propane and water. be sure your equation is balanced.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 10:30, kluckey3426
Asample of air with a volume of 2.20m3 at a pressure of 105 kpa and a temperature of 30c is cooled to 10c and the pressure is reduced to 75.0 kpa. what is the new volume? 6.9 1.34 2.56 43.0 2.88
Answers: 1

Chemistry, 22.06.2019 14:30, joejoefofana
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀ pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4. 0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3
You know the right answer?
Propane can be turned into hydrogen by the two-step reforming process. in the first step, propane an...
Questions in other subjects:







Mathematics, 24.07.2019 06:30


Health, 24.07.2019 06:30

Social Studies, 24.07.2019 06:30

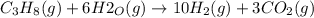
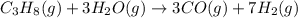 ...[1]
...[1]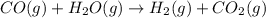 ...[2]
...[2]