
Chemistry, 27.09.2019 22:30 edwrads4044
Asap !
acetic acid has the following chemical formula:
ch3cooh
how many atoms of the element carbon (c) are in a molecule of acetic acid?
group of answer choices
3
2
1
4

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:30, jusicca1109
100 points answer quick the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1


Chemistry, 22.06.2019 18:40, bananaslada
What is the binding energy of a nucleus that has a mass defect of 5.81*10-^29 kg a 5.23*10-^12 j b 3.15* 10^12 j c 1.57*10-3 j d 9.44*10^20 j
Answers: 1

Chemistry, 22.06.2019 21:00, itasykamila
How many neutrons does an element have if its atomic number is 50 and its mass number is 166
Answers: 1
You know the right answer?
Asap !
acetic acid has the following chemical formula:
ch3cooh
...
acetic acid has the following chemical formula:
ch3cooh
...
Questions in other subjects:




Mathematics, 14.07.2020 21:01






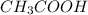 . And, symbol which represents a carbon atom is C, hydrogen atom is represented by H and an oxygen atom is represented by O.
. And, symbol which represents a carbon atom is C, hydrogen atom is represented by H and an oxygen atom is represented by O.


