

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, ksalinas7404
Un cierto gas tiene un volumen de 800ml a 80°c y 600ml a 80°c y 600mmhg de presión. ¿cual será el volumen del gas a condiciones normales? sí el gas es oxígeno, ¿cuál será su peso? y ¿cuántas moléculas están presentes en el sistema?
Answers: 2


Chemistry, 22.06.2019 22:30, eduardoguizar8787
Which one of the following bonds would you expect to be the most polar? a) b–h b) n–h c) p–h d) al–h e) c–h
Answers: 1

Chemistry, 23.06.2019 01:30, sheldonwaid4278
Magnesium is the limiting reactant in this experiment. calculate the theoretical yield of mgo for each trial. trial 1: trial 2: data mass of empty crucible with lid trial 1: 26.688 trial 2: 26.681 mass of mg metal, crucible, and lid trial 1: 26.994 trial: 2 26.985 mass of mgo, crucible, and lid trial 1: 27.188 trial 2: 27.180
Answers: 1
You know the right answer?
boyle's law represents the relationship of volume as pressure changes. the constants of boyle's law...
Questions in other subjects:

English, 10.12.2020 09:20

English, 10.12.2020 09:20

Mathematics, 10.12.2020 09:20


Chemistry, 10.12.2020 09:20

Chemistry, 10.12.2020 09:20

Mathematics, 10.12.2020 09:20

English, 10.12.2020 09:20


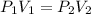 (T and n are constants)
(T and n are constants)
 and
and 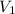 are the Initial pressure and volume and
are the Initial pressure and volume and
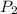 and
and 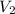 are the final pressure and volume
are the final pressure and volume


