
Chemistry, 25.09.2019 02:20 lexizamora2
1.00 kg of ice at -10 °c is heated using a bunsen burner flame until all the ice melts and the temperature reaches 95 °c. a) how much energy in kj is required to effect this transformation?

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 02:50, Jerrikasmith28
The conventional equilibrium constant expression (kc) for the system below is: 2icl(s) ⇄ i2(s) + cl2(g) [cl2] ([i2] + [cl2])/2[icl] [i2][cl2]/[icl]2 none of the listed answers are correct [i2][cl2]/2[icl]
Answers: 2


Chemistry, 22.06.2019 17:10, hahahwha
Acalorimeter is to be calibrated: 51.203 g of water at 55.2 degree c is added to a calorimeter containing 49.783 g of water at 23.5c. after stirring and waiting for the system to equilibrate, the final temperature reached is 37.6 degree c. specific heat capacity of water (s = 4.18 j/g∙degree c). calculate the calorimeter constant. (smδt)warm water = -[(smδt)cold water + (calorimeterδtcold water)]
Answers: 2
You know the right answer?
1.00 kg of ice at -10 °c is heated using a bunsen burner flame until all the ice melts and the tempe...
Questions in other subjects:





Mathematics, 21.01.2021 23:00



Physics, 21.01.2021 23:00


Biology, 21.01.2021 23:00

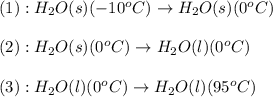
![\Delta H=[m\times c_{p,s}\times (T_{final}-T_{initial})]+n\times \Delta H_{fusion}+[m\times c_{p,l}\times (T_{final}-T_{initial})]](/tpl/images/0260/0438/5cd06.png)
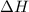 = energy required = ?
= energy required = ?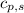 = specific heat of solid water =
= specific heat of solid water = 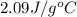
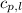 = specific heat of liquid water =
= specific heat of liquid water = 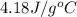
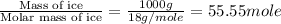
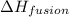 = enthalpy change for fusion = 6.01 KJ/mole = 6010 J/mole
= enthalpy change for fusion = 6.01 KJ/mole = 6010 J/mole![\Delta H=[1000g\times 4.18J/gK\times (0-(-10))^oC]+55.55mole\times 6010J/mole+[1000g\times 2.09J/gK\times (95-0)^oC]](/tpl/images/0260/0438/a7596.png)
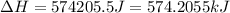 (1 KJ = 1000 J)
(1 KJ = 1000 J)

