
Chemistry, 25.09.2019 01:00 nene3210204
Calculate the pressure exerted by ar for a molar volume 0.45 l at 200 k using the van der waals equation of state. the van der waals parameters a and b for ar are 1.355 bar dm mol-2 and 0.0320 dm3mol? , respectively. write your answer (unit: bar) with 2 decimals, as 12.23. do not add unit to your answer.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:00, miamassimino
The rules of engagement (roe) working group is often used to (select all that apply.)
Answers: 2

Chemistry, 22.06.2019 04:40, marknjenbennetp3j1v1
Listen base your answer to the question on the information below. propane is a fuel that is sold in rigid, pressurized cylinders. most of the propane in a cylinder is liquid, with gas in the space above the liquid level. when propane is released from the cylinder, the propane leaves the cylinder as a gas. propane gas is used as a fuel by mixing it with oxygen in the air and igniting the mixture, as represented by the balanced equation below. c3h8(g) + 5o2(g) → 3co2(g) + 4h2o() + 2219.2 kja small amount of methanethiol, which has a distinct odor, is added to the propane to consumers detect a propane leak. in methanethiol, the odor is caused by the thiol functional group (–sh). methanethiol, ch3sh, has a structure that is very similar to the structure of methanol. what is the correct structural formula for a molecule of methanethiol
Answers: 3

Chemistry, 22.06.2019 08:30, dyanaycooper13
Analyze how limestone is weathered and identify the features that are formed as a result of this dissolution
Answers: 1

Chemistry, 22.06.2019 11:40, arlabbe0606
Effect of rotenone and antimycin a on electron transfer rotenone, a toxic natural product from plants, strongly inhibits nadh dehydrogenase of insect and fish mitochondria. antimycin a, a toxic antibiotic, strongly inhibits the oxidation of ubiquinol. (a) explain why rotenone ingestion is lethal to some insect and fish species. (b) explain why antimycin a is a poison. (c) given that rotenone and antimycin a are equally effective in blocking their respective sites in the electron-transfer chain, which would be a more potent poison? explain.
Answers: 3
You know the right answer?
Calculate the pressure exerted by ar for a molar volume 0.45 l at 200 k using the van der waals equa...
Questions in other subjects:

Chemistry, 19.02.2021 21:20

Mathematics, 19.02.2021 21:20

Mathematics, 19.02.2021 21:20

Mathematics, 19.02.2021 21:20

Mathematics, 19.02.2021 21:20


Mathematics, 19.02.2021 21:20



Biology, 19.02.2021 21:20

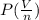 = RT
= RT 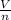 = v; which is called molar volume
= v; which is called molar volume = RT
= RT
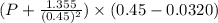 = 0.08314
= 0.08314 



